JINSP RS1000-DI RAMAN handled spectrometer
JINSP RS1000-DI RAMAN handled spectrometer
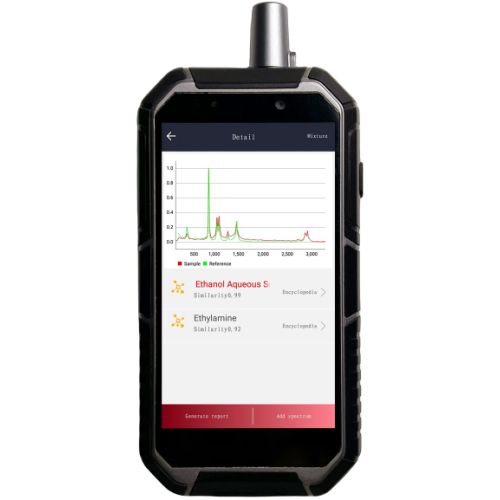
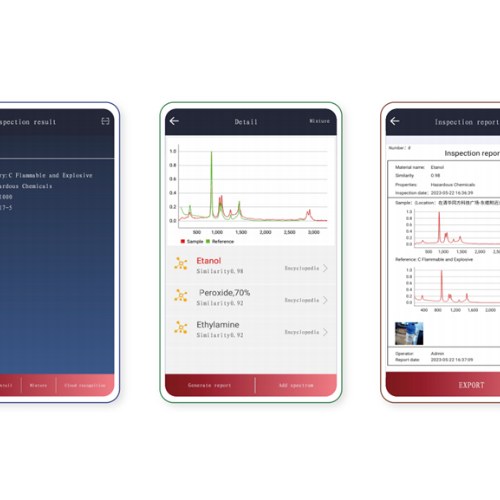
The RS1000-DI is a portable Raman spectrometer designed for the identification of pharmaceutical raw materials, featuring non-destructive identification capability: direct detection through glass packaging, woven bags, plastic, and paper — with no need to open containers or collect samples.
Applicable Regulations:
> GMP 2015, Chapter 6, Article 110: Appropriate standard operating procedures shall be established to confirm that raw materials and excipients in each container conform to specifications.
> Chinese Pharmacopoeia 2020, Chapter 0421: Raman spectroscopy may be used for identification testing and structural analysis.
> Pharmaceutical Inspection Co-operation Scheme (PIC/S): The identity of an entire batch can only be confirmed following identity testing performed on each individual container.
> FDA 21 CFR Part 11
> GMP 2015, Chapter 6, Article 110: Appropriate standard operating procedures shall be established to confirm that raw materials and excipients in each container conform to specifications.
> Chinese Pharmacopoeia 2020, Chapter 0421: Raman spectroscopy may be used for identification testing and structural analysis.
> Pharmaceutical Inspection Co-operation Scheme (PIC/S): The identity of an entire batch can only be confirmed following identity testing performed on each individual container.
> FDA 21 CFR Part 11
Labtech
Route Z4 - Z.I. Saint Gobain
1009 Jebel Jeloud
Tunisia
Copyright © 2026 Labtech | All rights reserved